Conceptualizing an ideal gas involves both the benchtop level perspectives expressed in the gas laws, which describe the interrelationship of macrostate functions. It also involves the particle-level perspectives of kinetic theory, which is the study of the microscopic behavior of molecules and the interactions which lead to macroscopic relationships in the gas laws.
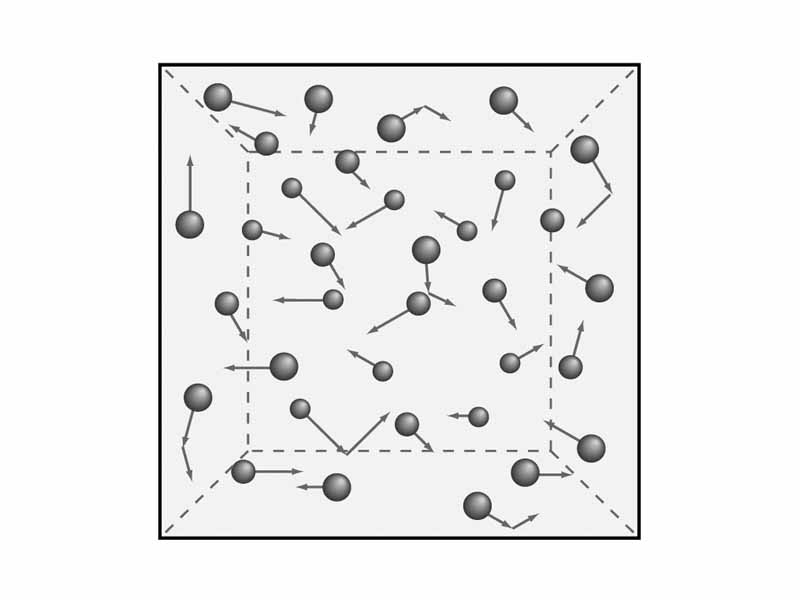
The ideal gas is an imaginary construct. Like the incompressible fluid or the frictionless inclined plane, the ideal gas was developed to make certain kinds of discussion possible. The ideal gas enables the discussion of basic thermodynamic relationships without the need for endless reservations. An ideal gas consists of perfectly spherical particles of zero volume that may only collide elastically. Because the particles may only interact through elastic collisions, as a system, an ideal gas may only possess internal energy in the form of the translational kinetic energy of the particles. There are no places for rotational or vibrational kinetic energy, and there are no intermolecular forces within an ideal gas. There is no electrostatic potential energy. One very important consequence of this restriction is that the internal energy of a given amount of ideal gas may be directly determined from the temperature. Another crucial consequence is that tthe thermodynamic state of the ideal gas may be specified by any two of the pressure, volume, and temperature.
In my opinion a good understanding of Thermodynamics is one of the most important steps to an integrated conceptual understanding of science, so the importance of this chapter, as well as all of the other topics from this week and next week cannot be understated. In addition the importance of the Ideal Gas and Kinetic Theory to the overall learning progression, this material will show up in conceptual questions and quantitative questions. Generally, the writers of the MCAT are more interested in your conceptual understanding of physics and chemistry than your ability to solve number problems, so the balance with MCAT physical sciences questions will lean more towards conceptual questions than quantitative questions. However, usually there will be a half dozen or so physics questions that require quantitative problem solving, so it is important to give a bit of extra attention to the categories that have often given rise to quantitative questions. On many MCATs there have been quantitative problems involving the Ideal Gas Law as well as portions of Kinetic Theory such as Graham's Law.
WikiPremed Resources
Ideal Gas & Kinetic Theory Cards
Conceptual Vocabulary Self-Test
Basic Terms Crossword Puzzle
Basic Puzzle Solution
Conceptual Vocabulary for The Ideal Gas
The Ideal Gas
The ideal gas law is the equation of state of a hypothetical gas consisting of identical particles of zero volume, with no intermolecular forces.
An ideal gas is a hypothetical gas consisting of identical particles of zero volume with no intermolecular forces undergoing perfectly elastic collisions.
Kinetic theory of gases attempts to explain macroscopic properties of gases by considering their molecular composition and motion.
Gas is one of the four major states of matter, consisting of freely moving atoms or molecules without a definite shape.
Boyle's law states that the product of the pressure and volume for an enclosed ideal gas will be constant if temperature remains fixed.
Charles's law states that at constant pressure, the volume of a given mass of an ideal gas increases or decreases by the same factor as its kelvin temperature increases or decreases.
Each gas in a mixture of ideal gases has a partial pressure which is the pressure which the gas would have if it alone occupied the volume.
The gas constant is a physical constant used in equations of state. It is another name for the Boltzmann constant, though expressed in units of energy per kelvin per mole rather than energy per kelvin per particle.
Graham's law of effusion states that the rate of effusion of a gas is inversely proportional to the square root of the mass of its particles.
Effusion is the process where individual molecules flow through a small pore without collisions.
Dalton's law of partial pressures states that the total pressure exerted by a gaseous mixture is equal to the sum of the partial pressures of each component in a gas mixture.
The van der Waals equation is the general equation of state for a fluid composed of particles that have a non-zero size and a pairwise attractive inter-particle force.
Gay-Lussac's law states that the ratio between the combining volumes of reagent gases and product can be expressed in small whole numbers.
The Boltzmann constant is the physical constant relating temperature to energy.
In statistical mechanics, a degree of freedom is a single scalar number describing the classical micro-state of a system.
Statistical mechanics is the application of probability theory to the field of mechanics.
The equipartition theorem, which relates the temperature of a system to its average energies, depends on the idea that at thermal equilibrium, energy is shared equally among its various forms within the system.
Root mean square speed is the measure of the speed of particles in a gas that is most convenient for problem solving within the kinetic theory of gases.
The Boltzmann distribution predicts the distribution function for the fractional number of particles occupying a set of states depending on the temperature and degeneracy of the system.
Ludwig Eduard Boltzmann (1844 - 1906) was an Austrian physicist famous for his founding contributions in the fields of statistical mechanics and statistical thermodynamics.
In statistical mechanics, the ensemble average is defined as the mean of a quantity that is a function of the distribution of micro-states of a system.
A departure function for a species is the difference between a property as computed for an ideal gas and the property as it exists in the real world, for a specified temperature T and pressure P.
An ideal Bose gas is a quantum-mechanical version of a classical ideal gas. It is composed of bosons, which have an integral value of spin.
The virial theorem provides a general equation relating the average total kinetic energy of a thermodynamic system with its average total potential energy.

