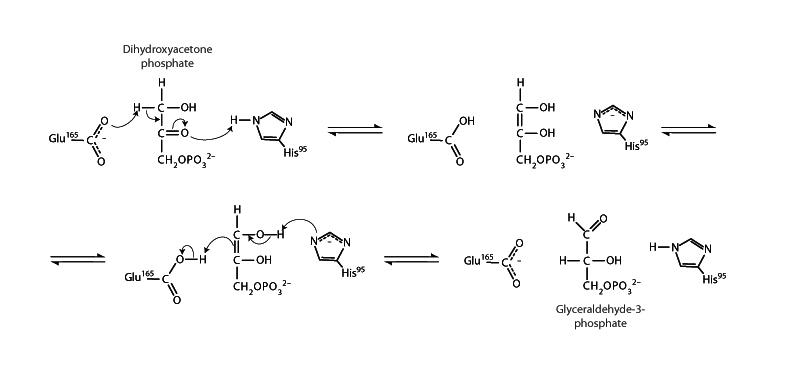
Triose phosphate isomerase (TPI) plays an important role in glycolysis and gluconeogenesis catalyzing the reversible interconversion of the ketose dihydroxyacetone phosphate (DHAP) with its aldose isomer glyceraldehyde 3-phosphate (GAP). The mechanism provides an interesting example of keto-enol tautomerism. The interconversion of DHAP and GAP proceeds through an intermediate that is an enol tautomer of either triose. Because there are two hydroxyl groups to choose from in returning to the carbonyl form, keto-enol tautomerism can lead back and forth between the two sugars.
One interesting feature of the TPI reaction is that DHAP is actually the thermodynamically favored isomer, representing 96% of the reaction mixture at equilibrium, though GAP represents the substrate
of the next step in glycolysis, the glyceraldehyde 3-phosphate dehydrogenase step. Even though formation of DHAP is thermodynamically favored, the reaction readily moves forward in glycolysis. Why is that? This is due to the continuous removal of GAP by glyceraldehyde 3-phosphate dehydrogenase. In other words, the triose phosphate isomerase reaction moves forward by mass action. LeChatelier's principle tells us that removal of the product will shift the reaction toward formation of more product.