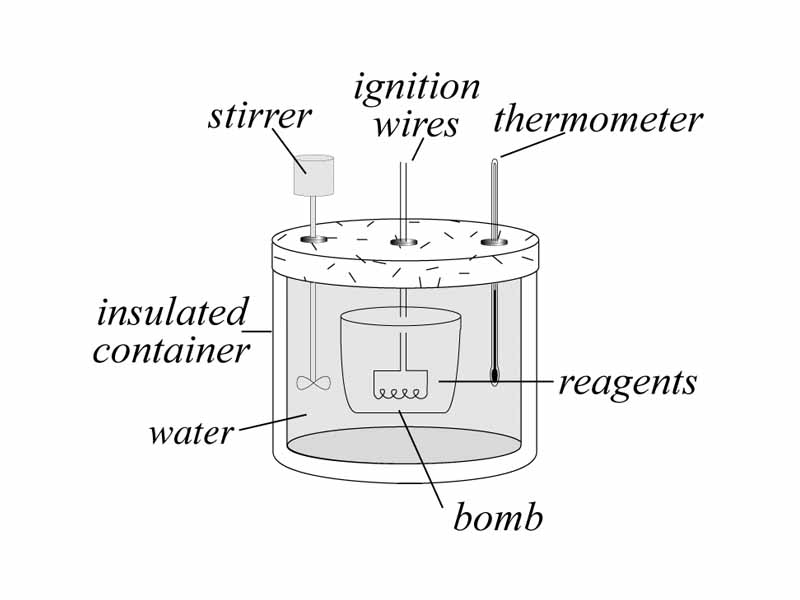
Thermochemistry is the application of the 1st law of thermodynamics to chemical transformations. Thermochemistry is conservation of energy applied to chemistry. In both topics, work means the the product of the pressure and the change in volume of the system. In addition to thermodynamic work, heat flow is the other way that physical and chemical systems can exchange energy with their surroundings. However, thermochemistry defines a state function of the system whose change will equal the heat flow (as long as constant pressure is maintained. This state function is the enthalpy. When you define things so that heat flow equals the change in enthalpy it allows heat flows to be summed over alternative pathways. Heat flow can be described in a way that is path independent. This step is a foundation of chemical reasoning. The formalism that makes this possible in chemistry is to assume that pressure is constant over the path. In that case, the change in the enthalpy, ie. the change in the internal energy plus the pressure volume product, will exactly equal equal heat flow.
One important thing to consider when applying the 1st law to chemistry is that internal energy is a much richer idea in chemistry than in the simple model system of physics, the ideal gas in a piston. In physics, we kept pretty much to the ideal gas whose internal energy is exactly and only the kinetic energy of the particles. In chemistry, though, the internal energy is much more complicated. It helps to understand that there really are two primary kinds of internal energy relevant to chemistry. These are the kinetic energy, which will have rotational and vibrational modes in addition to translational modes in most chemical substances, and the electrostatic potential energy associated with the arrangement of charged particles at the atomic, chemical bonding, and intermolecular levels. Crucial to being able to conceptualize chemical change in the context of the 1st law of thermodynamics is to have a clear sense of the electrostatic potential energy changes in chemical substances as they impact internal energy change. Which bonds are broken? Which bonds are formed?