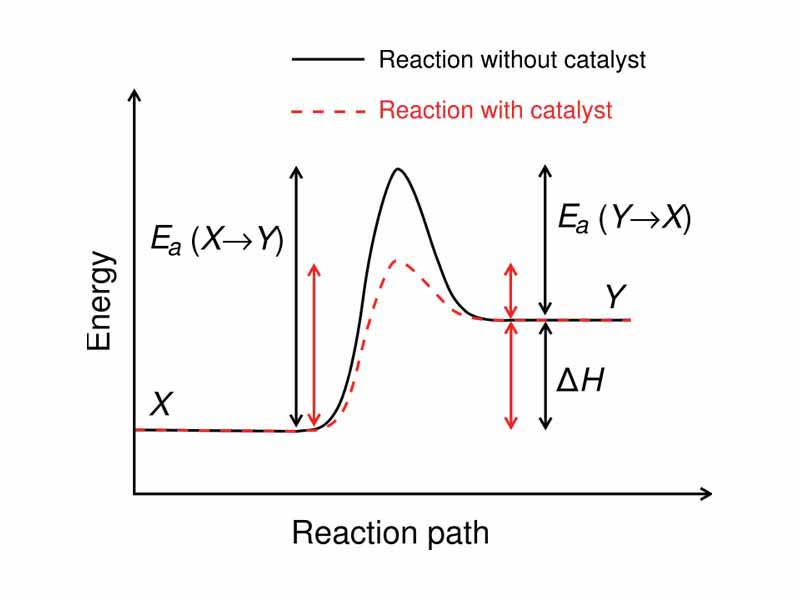
A reaction rate is the speed at which reagents are consumed and products are created. Chemical Kinetics is the study of the rate of chemical reactions. Because the rate of a chemical reaction depends on its pathway, different kinetics may apply for the same overall transformation if it can occur through a variety of mechanisms. Path dependence fundamentally distinguishes the propositions of Chemical Kinetics from those of Chemical Thermodynamics. Chemical Thermodynamics depends solely on the comparison of the initial and final state of the system and surroundings, without recourse to discussion of the reaction pathway, unlike Chemical Kinetics. The use of a catalyst, for example, will change the kinetics of a reaction, but it will not change the position of equilibrium, which is a thermodynamic function.
Chemical Kinetics problems involving number crunching are rare on the MCAT. However, conceptual questions involving Chemical Kinetics are very common on the test. Chemical kinetics lends itself especially well to the MCAT passage format where passages presenting data can lead to questions involving inference of reaction order, for example, or where the application of kinetics principles allows one to judge among various reaction mechanisms. In the construction of multiple choice questions, the MCAT writers are also fond of questions that make sure students don't confuse the propositions of Chemical Kinetics, which depend on the reaction pathway, with the concepts of Chemical Thermodynamics, which do not depend on the mechanism.
WikiPremed Resources
Chemical Kinetics Practice Items
Conceptual Vocabulary Self-Test
Basic Terms Crossword Puzzle
Basic Puzzle Solution
Conceptual Vocabulary for Chemical Kinetics
Chemical Kinetics
Chemical kinetics is the study of reaction rates in a chemical reaction.
The reaction rate for a reactant or product in a particular reaction tells you how fast a reaction takes place.
Catalysis is the increase in rate of a chemical reaction by means of a substance called a catalyst.
The order of reaction with respect to a certain reactant is defined, in chemical kinetics, as the power to which its concentration term in the rate equation is raised.
The rate-determining step is a chemistry term for the slowest step in a chemical reaction.
An intermediate in a chemical reaction is a molecular entity with a lifetime appreciably longer than a molecular vibration that is formed from the reactants and reacts further to give the products of a chemical reaction.
A reaction mechanism is the step by step sequence of elementary reactions by which overall chemical change occurs.
A reaction coordinate is an abstract one-dimensional trajectory representing progress along a reaction pathway.
The transition state of a chemical reaction is a particular configuration along the reaction coordinate defined as the state corresponding to the highest energy along this reaction coordinate.
An activated complex is a transitional structure in a chemical reaction that results from an effective collision between molecules and that persists while old bonds are breaking and new bonds are forming.
Activation energy, also called threshold energy, is a term defined as the energy that must be overcome in order for a chemical reaction to occur.
The rate law or rate equation for a chemical reaction is an equation which links the reaction rate with concentrations or pressures of reactants and constant parameters.
Transition state theory is a conception of chemical reactions or other processes involving rearrangement of matter as proceeding through a continuous change in the relative positions and potential energies of the constituent atoms and molecules.
A reaction step of a chemical reaction is defined as an elementary reaction, constituting one of the stages of a reaction in which a reaction intermediate is converted into the next reaction intermediate in the sequence between reactants and products.
Homogeneous catalysis describes catalysis where the catalyst is in the same phase as the reactants.
Heterogeneous catalysis describes catalysis where the catalyst is in a different phase to the reactants.
An elementary reaction is a chemical reaction in which one or more chemical species react directly to form products in a single reaction step and with a single transition state.
A single chemical reaction is said to have undergone autocatalysis if the reaction product is itself the catalyst for that reaction.
Hammond's postulate states that if a transition state and intermediate occur consecutively during a reaction process and have nearly the same energy content, their interconversion will involve only a small reorganization of the molecular structures.
Molecularity in chemistry is the number of colliding molecular entities that are involved in a single reaction step.
The Arrhenius equation is a simple, but remarkably accurate, formula for the temperature dependence of a chemical reaction rate, more correctly, of a rate coefficient.
A reaction inhibitor is a substance that decreases the rate of, or prevents, a chemical reaction.
A catalytic cycle in chemistry is a term for a multistep reaction mechanism that involves a catalyst.
Collision theory assumes that for a reaction to occur the reactant particles must collide, but only a certain fraction of the total collisions, the effective collisions, cause the transformation of reactant molecules into products.
Biocatalysis can be defined as the utilization of natural catalysts, such as protein enzymes, to perform chemical transformations on organic compounds.
A potential energy surface is a representation of interactions in simple chemical and physical systems in which the total energy of an atom arrangement can be represented as multidimensional surface with atomic positions as variables.
A steady state is a situation in which all state variables are constant in spite of ongoing processes that strive to change them.
The katal is the SI unit of catalytic activity.
The Eyring equation in chemical kinetics, which relates the reaction rate to temperature, follows from transition state theory, contrary to the Arrhenius equation, which is empirically based.
A temperature jump is a piece of apparatus useful in the study of chemical kinetics, involving the discharging of a capacitor through a small volume solution containing the reaction to be studied.